Substance use disorders/Expert
The following article deals with sex/gender differences in substance use disorders.
Contents
Epidemiology
Incidence/Prevalence
According to a study from 2013, 31.2 percent of men and 15.2 percent of women in Germany have been addicted to some substance at least once in their lives.[1] The significantly higher prevalence of males who have ever been affected results from various current surveys: For most substances, the prevalence of general, risky or dependence-induced use is higher among men than among women, although for some substances there are converging trends.[2] The higher prevalence of the male sex with regard to most substances usually only manifests itself in cohorts of young adulthood. In addition, men usually consume a larger amount of the same substance. Thus, gender differences are most evident in the area of the highest reported consumption. Nevertheless, there are indications that the prevalences of both sexes (especially for smoking) converge, i.e. in younger cohorts the gender differences are lowest, in older cohorts highest. It can be assumed that these cohort effects reflect a cultural change. While smoking was not well regarded among women in the past, it is socially accepted today. It needs to be examined whether this is really a cultural phenomenon or whether the differences in the cohorts represent stable values over different life stages. Further research is needed in this regard.[3]
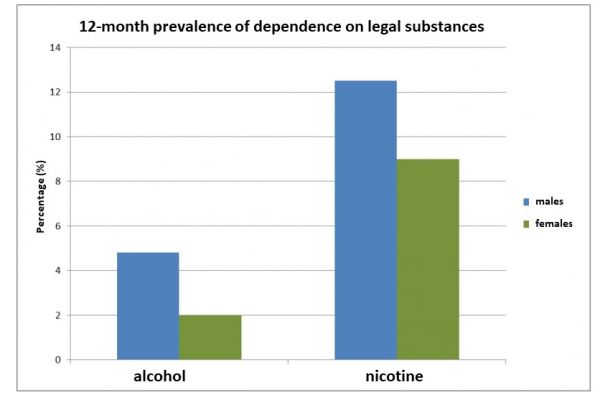
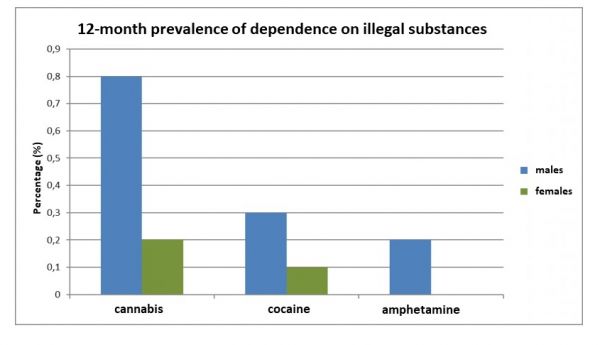
The 12-month prevalences of dependence (according to DSM-IV) on licit and illicit substances can be found in figures 1 and 2.
Substance-specific epidemiological data can be found under the following links:
Figure 1. 12-month prevalence of dependence on alcohol and nicotine; n = 9084, age: 18-64 [Source: GenderMed-Wiki, according to Pabst et al. (2013)].
Figure 2. 12-month prevalence of dependence on cannabis, cocaine and amphetamines; n = 9084, age: 18-64 [Source: GenderMed-Wiki, according to Pabst et al. (2013)].
Amphetamines
Based on a survey by Papst et al. (2013), more men than women have used amphetamines in Germany in a one-year period (12-month prevalence of 1.2 percent versus 0.3 percent, self-reported). Among men, the prevalence of amphetamine abuse is about 0.3 percent and for amphetamine dependence 0.2 percent (according to DSM-IV), among women neither abuse nor dependence could be identified. Looking at age groups, the prevalence of amphetamine abuse does not follow any trend (neither increasing nor decreasing with age). Most persons with amphetamine dependence were in the age group of 25 to 29 years (one percent), followed by 18 to 20 years (0.4 percent) and 21 to 24 years (0.1 percent). No dependencies could be identified in all other age groups.[2]
Medication
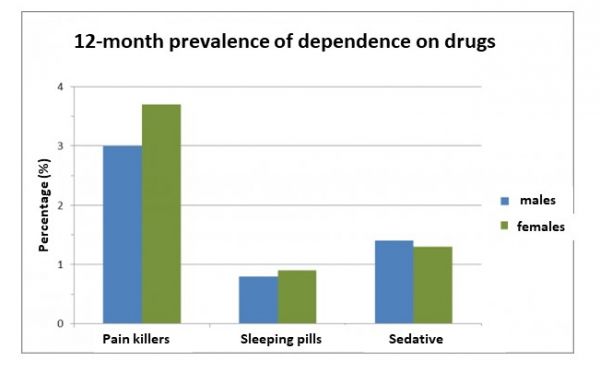
The sex-specific 12-month prevalences of dependence on painkillers, sleeping pills and tranquillisers are shown in figure 3. It is important to note that medication is taken less for pleasure and intoxication than primarily for its therapeutic effect and according to a doctor's prescription. A clear gender difference is only found with regard to dependence on painkillers (higher prevalence among women).
Figure 3. 12-month prevalence of drug dependence; n= 9084, age: 18-64 [Source: GenderMed-Wiki, according to Pabst et al. (2013)].
Painkillers
In Germany, more women than men take painkillers within one year (68 percent versus 56.1 percent) and also consume them more often on a daily basis (one-month prevalence of 4.6 percent versus 3.8 percent). The DSM-IV criteria for abuse (8.8 percent versus 8.5 percent) and dependence (3.7 percent versus 3 percent) were also met more often in women than in men.[2]
Sleeping pills
The 12-month prevalence of sleeping pill use in Germany is higher among women (6.8 percent) than among men (4.2 percent). The rate of daily consumption is slightly higher for women (0.8 percent) than for men (0.7 percent). With regard to sleeping pill abuse (according to DSM-IV), there is no gender difference (0.8 percent each). In contrast, women with 0.9 percent suffer from sleeping pill dependence (also according to DSM-IV) slightly more often than men with 0.8 percent. Frequency of use as well as daily use tends to increase across age groups, while abuse and dependence are relatively evenly distributed across them.[2]
Tranquillisers
In Germany, 6.2 percent of women, but only 4.6 percent of men take some kind of tranquilliser within a year. For women, there is also a slightly higher daily intake (1.3 percent versus 1.1 percent). There are no gender differences regarding abuse (0.8 percent each, according to DSM-IV), but men are somewhat more often dependent (1.4 percent versus 1.3 percent, according to DSM-IV). For all the above-mentioned intake characteristics, there is a trend towards higher values with age.[2]
Other drugs
Stimulants (psychostimulants, e.g. for ADHD or narcolepsy) are more frequently used by men in Germany (12-month prevalence of 1.2 percent versus 0.7 percent; daily 0.3 percent versus 0.2 percent). Use took place especially in younger to middle cohorts (18 to 39 years). Women in Germany are more likely to use appetite suppressants within a year (0.6 percent versus 0.2 percent), but men are more likely to do so daily (0.2 percent versus 0.1 percent). Use is most prevalent in younger to middle-aged cohorts.[2]
Substances in brief
The 12-month prevalence of use of the following (illicit) substances was higher among men than among women in Germany: Ecstasy (0.7 percent versus 0.1 percent), LSD (0.5 percent versus 0.1 percent), heroin (0.3 percent versus 0.1 pe cent), crack (0.2 percent versus <0.0 percent), mushrooms (0.5 percent versus. 0.1 percent), spice (0.3 percent versus <0.0 percent) and other opiates (0.4 percent versus 0.3 percent).[2]
Risk and protective factors
Most risk factors for the development of a dependence disease do not differ between men and women. Nevertheless, there are also some gender-specific risk factors, which are explained below.
Impulsivity, sensation seeking, self-regulation
Impulsivity, sensation seeking and self-regulation are considered essential predictors for the development of addiction, which are shared by both sexes.[3]
Importantly, inhibitory control and substance use influence each other, and substance use in turn weakens the ability to control behaviour.[4] Neurobiological developments occur during puberty that promote tendencies for sensation seeking at a time when impulse control is still developing.[5]
Sensation seeking and impulse control scores differ between males and females and are related to pubertal development: although both sexes achieve elevated sensation seeking scores during puberty, adolescent males generally show higher levels of sensation seeking and lower levels of impulse control. In general, it can be stated that the gender difference in both personality traits increases with age and that boys in particular therefore become vulnerable to risky behaviour (e.g. excessive substance use) during adolescence. However, puberty typically begins earlier in girls, so that they develop elevated levels of sensation seeking at a younger age due to the associated changes, and become vulnerable to substance use earlier than boys.[5]
Impulsivity is often divided into two variants:[6] Impulsive choice describes behaviour that occurs without concern for future consequences, which is usually demonstrated in experimental studies by a preference for a small but immediate reward rather than a larger but later reward. Impulsive action, on the other hand, describes the inability to withhold a reaction until a certain time is signalled (e.g. operationalised by a stop signal, after the presentation of which no button to be pressed beforehand should be pressed again).
While both variants of impulsivity are associated with substance abuse, higher levels of the impulsivity variant impulsive choice in particular correlate with diagnosed dependence.[7] Although there are only a few studies in which the impulsivity variant impulsive choice has been investigated in animals, these indicate moderately higher scores in favour of females.[8] Gender-specific differences with regard to the impulsivity variant impulsive choice can be found in the study.
Gender differences regarding the expression of the impulsivity variant impulsive choice are less clear in humans; consistent results do not exist. Nevertheless, some trends have been found:[8] With regard to impulsive choice, females show higher scores for hypothetical rewards, whereas males show higher scores for actual rewards. Regarding the impulsivity variant impulsive action, male laboratory animals show higher scores, especially when sex hormones are taken into account, while differences in humans are moderate and task-specific (i.e., males are more impulsive in continuous performance tasks and go/no-go tasks, and females are more impulsive in stop-signal tasks).[9] The results of these studies have shown that males are more impulsive than females in continuous performance tasks and go/no-go tasks.
Women have been shown in some studies to act more impulsively than men during substance use. In contrast, in the control groups (i.e. without using), male subjects act just as impulsively or more impulsively than female subjects. It can be concluded that there is a gender-specific covariance between impulsive behaviour and substance abuse.[6] Impulsive behaviour in women is also cycle-dependent:[10] women are least impulsive during the follicular phase, which may also explain crucial differences between studies. In addition, the gender-specific expression of impulse control can possibly be attributed to differences in the orbitofrontal cortex (OFC), whose effect on impulsive decisions is well documented:[4] The OFC is larger in women, has a lower glucose consumption, the receptors for oestrogens and androgens are more densely populated there and the connectivity with the prefrontal cortex and the dorsal striatum is higher. In contrast, the volume of the prefrontal cortex is lower in young female alcohol consumers compared to young alcohol consumers and same-sex controls (both 15 to 17 years). It can be concluded that biological sex may modulate the influence of substances on the morphology and activation of cortical areas associated with impulsivity.[11]
Mesolimbic-System
The mesolimbic system with its dopaminergic pathways plays a crucial role in the processes underlying a dependence syndrome.[4] In this context, altered functioning of areas in the mesolimbic system could be both one of the causes for the development of dependence and a consequence of it. Gender differences are found in animal studies with regard to dopamine concentration in the striatum, among other things, where oestrogens had a gender-specific effect on the binding of dopaminergic D2 receptors in female animals.[12] In rodents, gender-related differences are also found in the properties and plasticity of mesolimbic dopaminergic neurons. These differences may be one reason why women learn substance use more quickly than men.[13]
Beginning of puberty and its course
During puberty, developmental processes take place on a biological, social and environmental level, the interactions of which make it difficult to point out clear correlations. The age of onset of puberty, elevated scores in personality traits such as sensation seeking and impulsivity as well as comorbidities (e.g. ADHD, depression) are considered important risk factors in adolescents of both sexes. Nevertheless, there are gender-specific differences in the effect sizes of all factors, which develop during puberty and are usually most pronounced in adulthood.[3] Thus, these changes can only be understood in a temporal, developmental context and are described individually below.
In a longitudinal study, adolescents who reported being further along in pubertal development than their peers were more likely to have used cigarettes, alcohol and marijuana in the last three months.[14] Since girls reach puberty earlier (and there are clear correlations between substance use and self-reported pubertal development as early as age eleven), girls may be more vulnerable to first use during puberty than boys.
Sensation seeking, which is generally higher in boys and men, is often mentioned as a mediator variable for the connection between pubertal development and early substance use or abuse. In girls, there is an increased incidence of depression and anxiety during puberty. These conditions correlate with (increased) substance use.[3]
Overall, puberty turns out to be a critical phase, as on the one hand adolescents are more receptive to reward effects of substances, and on the other hand aversive effects (e.g. withdrawal symptoms) are felt less strongly.[6] Both characteristics can lead to a positive bias towards the drug experience. To make matters worse, many changes take place at the neural level during puberty, so that increased substance use in adolescence can have serious neurological consequences. For example, functionality in the prefrontal cortex may change in such a way that the user's impulsive behaviour increases (weakened top-down control), which could then be a risk factor for substance abuse.[6]
Comorbidities
Substance use disorder is often accompanied by a comorbid mental health disorder that can accelerate the progression of substance use.[3] These mental health disorders include anxiety, depression, bipolar disorder, conduct disorder and attention deficit hyperactivity disorder (ADHD). Gender differences, which usually develop during adolescence, exist for each of these disorders:[3]
Males develop comorbid conduct disorder and ADHD more often. In addition, these disorders are also considered risk factors for substance abuse in male adolescents. A high prevalence of conduct disorders has also been found in female adolescents with problematic substance use. Depression, anxiety and bipolar disorder are more often observed comorbidly in females. The consequence of these disorders is then often the consumption of alcohol to combat manic symptoms or to alleviate depressive symptoms. The comorbidity of dysthymia and double depression is the same between the sexes.
Traumatic experiences (especially during childhood) are associated with substance abuse. Significantly more women than men are victims of sexual abuse (prevalence in women c.a. 26.5 percent, in men c.a. 4 percent).[15] Furthermore, sexual abuse experiences are associated with problematic substance abuse only in women.
Another study examined the association between five forms of child maltreatment (physical, sexual and emotional abuse as well as physical and emotional neglect) and various substance use disorders,[16] finding that all five forms were associated with an increased likelihood of substance abuse in women, but that in men, the forms of physical and emotional neglect were not associated with abuse of certain substances (heroin, amphetamines and cocaine). The presence of child maltreatment thus proves to be a predictor of age of onset and severity of substance abuse in both sexes, but this is significantly more pronounced in females.[17]
Sex hormones
An isolated consideration of the influence of sex hormones on substance use is difficult, as there is always an interaction with relevant social and biological systems and developmental processes.[18]
Nevertheless, oestradiol has been found to increase both substance acquisition and the reward effect of the substance in female experimental animals and in female subjects, whereas this is not the case in male experimental animals and male subjects. [6][19] The female cycle forms an important influencing factor here: in the first phase of the cycle (follicular phase), oestradiol increases strongly, with which craving (desire for a certain substance) also becomes stronger, while in the second phase of the cycle (luteal phase) the increase in progesterone balances out this gender effect. In a similar way, chronic use of oral contraceptives, which act on the natural hormonal cycle, is a risk factor for women.[18]
With regard to male sex hormones, the testosterone concentration of both sexes in particular has been shown to be predictive of alcohol consumption.[20] It should be noted that alcohol consumption initially increases the testosterone concentration in both sexes, but chronic alcohol consumption in men then leads to a lower testosterone concentration, which is not observed in women. Studies suggest that the effect of testosterone on substance use is mediated by increased impulsive behaviour and/or sensation seeking.[3]
Social relationships
Drug use influences social behaviour, but social contacts (especially the peer group) also often influence drug use. That is, social contact with people who use drugs increases the likelihood of using drugs oneself.[3] The social environment can have a rewarding and reinforcing effect on drug use (social interaction is facilitated and group membership is increased), with processes of social learning (learning from the model) playing an important role. In the same way, a social environment that does not use drugs can have a preventive effect or reduce drug use.[21] In animal experiments with adolescent rats, it has been found for both sexes that alcohol, nicotine and cocaine can facilitate social interactions and thus promote social reward, which further increases substance use in the group.[3] However, gender can also influence use behaviour within the group or partnership. Results in animal studies suggest that the gender of the social partner can moderate the relationship between social interaction and alcohol consumption.[22] In future studies, the gender of social partners should therefore definitely be taken into account.
A drug-using peer group seems to increase the risk of starting drug use in boys earlier (at the age of ten to twelve years) than in girls (from 16 years onwards).[23] However, already from puberty onwards, it is shown that girls and women are influenced to a higher degree by the substance use (at least proven for alcohol and cigarettes) of their romantic partner than boys and men.[3]
Pathophysiology
It is important to note that the research is based on paradigms that use substance-naive animals, low to moderate doses and/or comparatively demanding schedules of reinforcement.
Sex differences are less likely to occur with high doses (or very easy access to the substance) because of what is known as a ceiling effect. In addition, in human studies, participants are usually not substance naïve, i.e. they usually already have experience with the use of the respective substance.
Although men use almost all substances more and have higher prevalences of substance use disorders, women develop dependence more quickly and go through typical phases of substance use earlier or faster (i.e. lower age of onset, earlier entry into rehabilitation, shorter duration of substance abstinence).[24] In animal studies, female rodents also start using substances more quickly and also become dependent earlier (e.g. on nicotine, cocaine, amphetamines and alcohol). In addition, there is evidence that increased consumption is associated with gender-specific neuroadaptations.[3] This sex difference is attributed to the fact that females are more susceptible to reward effects of psychoactive substances due to the activity of oestrogen.[25] Related to this, the subjective effect of substances in females varies depending on the menstrual cycle. I.e. in the follicular phase, the enhancement mechanisms are stronger in women due to the higher concentration of oestradiol, while progesterone has an inhibitory effect in the second phase.[26] In a fMRI study, it was confirmed that the reward system responds more strongly than usual to psychoactive substances during the mid-follicular phase.[27]
The finding that women have an accelerated progression from initial use to dependence and initiation of initial treatment has been demonstrated for opioid, cannabis and alcohol dependence and is referred to in the literature as the telescoping effect. [28] In addition, the telescoping effect is also used to describe the observation that women show stronger medical, behavioural or social problems than men at their first treatment, although they have used a certain substance on average less and for a shorter period of time.[28] This could be due to fundamental gender differences in the metabolisation of substances (see also Pharmacological principles).
Substance-specific data on pathophysiology can be found under the following links:
Clinical presentation
Symptoms
Due to the telescoping effect, women who seek treatment often show a higher dependence as well as a higher prevalence of comorbid mental disorders.[29] Both craving and withdrawal symptoms are more pronounced in women.[29] In addition, the triggers of craving may differ between the sexes. Reasons for nicotine or alcohol consumption are more often positive reinforcement and pleasure in men, while women consume more often as a coping strategy or for emotion regulation. In addition, smoking is more often triggered by cues in women.[19][3] Women with very high alcohol consumption also react more strongly to stress and social problems with craving and relapse into addiction, while men are more sensitive to substance-associated stimuli during stress (which then in turn promote craving and relapse into addiction). Behavioural analogies are also evident in animal studies with female and male rodents.[30]
Diagnostics
For comprehensive diagnostics, the following gender-specific aspects should be taken into account: Dependent women are less likely to seek specialised facilities, but are more likely to seek support in general medical practices, which seems to be related, among other things, to the social stigma that dependence is a "male disorder."[31]
Furthermore, as women are more likely to suffer from comorbid disorders (e.g. anxiety and depression), this increases the likelihood of a diagnosis in which addiction may not be properly considered. For example, a single diagnosis of depression is often made, with the person additionally trying to cope with their depression symptoms through substance abuse. The reverse is true for male patients: In psychiatry, the phenomenon of so-called depression blindness exists in men, which leads to the fact that despite the same symptom expression, women are more likely to be diagnosed with depression than men.[32] It can then happen that only a dependence disorder is recognised, while the comorbid depressive symptoms are neglected. Overall, more attention needs to be paid to both sexes with regard to psychiatric comorbidity, but also with regard to the incentive motivation to start therapy.
Patient management
Therapy
Physician-patient interaction
The attending physician must develop an understanding of the specific needs of women, in particular, as these can differ considerably from the typical characteristics of male patients. In fact, dependence is often seen as more severe in women because typical "male" dependence is used as a point of comparison.[31]
Women are also more likely to visit general practitioners in cases of dependence, which is why it is also important in this setting to record their exact physical and psychological condition and to prescribe therapies in a gender-adapted manner.[33] This includes, for example, addressing traumatic events (such as sexual abuse) - after which affirmation, placement in group therapies with men should be avoided - or also taking children into account, for whom women more often feel responsible and therefore drop out of therapy.
Treatment outcome
In their 2007 review, Greenfield et al. examined 280 studies and found that the variable gender was not a significant predictor of treatment duration, discontinuation or success. However, gender-specific predictors do exist, and various treatment elements can influence treatment success depending on gender. For example, better outcomes for female patients may be achieved by addressing problems that are most prevalent among affected women or tailored to different subgroups (e.g. older women). Across the lifespan, substance-dependent women are less likely to start treatment than affected men.[33] Different treatment options are explained in detail below.
Pharmacotherapy
Many pharmacotherapeutic concepts for the treatment of addictions are based on animal experiments. A relatively stable finding is that many substances work more effectively in female animals than in males. In humans, most substances so far either show no effect or have significant side effects.[6]
Nevertheless, there are some drugs that have been shown to be effective in reducing craving or absolute consumption. For many of these drugs it has turned out that there is a gender-specific effect (cf. Table 1). Nevertheless, it is currently not common in the field of pharmacological therapy to adjust drug doses to the respective gender. Nevertheless, there are sufficient indications (e.g. lower or no effects or stronger side effects) that women would benefit from dosages that take gender or at least body weight into account. [19]
Table 1. Sex differences in the effects of different drugs on substance use disorders.
|
Medication |
Sex-specific effect |
|
Naltrexone |
For naltrexone, it is unclear whether there are differences in effectiveness between the sexes.[34] Various studies show reduced consumption and craving for both sexes through the use of naltrexone (often in combination with psychological therapies).[35][36] As several substances are often consumed in parallel, interactions can occur. These interactions can be influenced by gender and should be taken into account during pharmacotherapy.[37] In a study with persons who were addicted to both alcohol and cocaine, only men reduced their substance consumption after administration of naltrexone, whereas women increased it.[38] The authors justified this with the rather high dose (150 mg/day), which may have triggered stronger side effects in women and thus impaired effectiveness. In line with this assumption, another study showed that side effects (such as nausea) were more frequent in women treated with naltrexone compared to men and controls.[39] In addition, only women showed increased cortisol levels and were more affected by side effects when they were in the luteal phase rather than the early follicular phase. So far, mainly the oral intake of naltrexone has been investigated. However, an injectable version of naltrexone also exists, the effectiveness of which seems to be more proven in alcoholic men.[40] |
|
Other medications for alcohol addiction & alcohol withdrawal syndrome |
For some medications used for the treatment of alcohol addiction or alcohol withdrawal syndrome, it remains unclear whether gender differences in effectiveness exist. Due to the insufficient number of women in clinical trials, no firm statements can be made about the gender-specific effectiveness and safety of disulfiram, anticonvulsants, gamma-hydroxybutyric acid and benzodiazepines. Only for nalmefene and acomprosate was the size of the female subject collective sufficient, so that here it could be established that there were no gender differences.[34] |
|
Fluoxetine |
Fluoxetine taken eight weeks before smoking cessation reduced depressive symptoms, craving and withdrawal-related negative affect only in women. For women, depressive symptoms occurring before smoking cessation and craving are associated with abstinence duration, whereas for men, withdrawal-related negative affect is associated with abstinence duration.[41] |
|
Pregabalin |
In direct comparison with a daily application of naltrexone (50 mg), pregabalin shows a greater effect in terms of a stronger reduction of alcohol consumption. This is possibly related to the anxiolytic effect of pregabalin, as comorbid psychiatric symptoms (e.g. anxiety) often accompany alcohol addiction. The study thus indicates that drug treatment is more successful if comorbid psychiatric disorders are taken into account in the process.[36] |
|
Nicotine replacement therapy |
Men seem to benefit more from nicotine patches or nicotine gum than women. More women than men discontinue treatment with nicotine patches due to skin irritation.[19] Overall, the evidence is inconclusive. |
|
Bupropion |
Bupropion appears to be more effective in maintaining abstinence from smoking in women. This may be related to the fact that it reduces weight gain during smoking cessation.[19] |
|
Methadone |
Men may be more susceptible to QT prolongation of the heartbeat at low doses of methadone, although the overall risk of developing QT prolongation is higher in women.[19] |
|
Buprenorphine |
After application of the same dose, women reach significantly higher plasma concentrations of buprenorphine. Compared to methadone, it is more effective in women, and it also seems to be possible to take it in pregnant women (due to a lower neonatal withdrawal syndrome, it is preferable to methadone).[19]. |
|
Progesterone |
Since progesterone can have a positive effect on substance abstinence, it was investigated whether exogenous administration also proves effective. In animal experiments, administered progesterone reduces cocaine use. A positive effect in the sense of reduced physiological and subjective reward effects and lower cravings can also be demonstrated in humans.[6] These effects are particularly evident in female animals or subjects. Cocaine use has also been shown to be reduced by progesterone administration in women who have just given birth.[42] In a study in which, however, only ten people participated, it was shown for both men and women that the subjective and physiological reaction to cocaine use is reduced after taking progesterone.[43] |
Wider therapeutic options
Table 2. Gender differences in non-drug treatment options.
|
Therapy approach/content |
Gender-sensitive consideration |
|
Consideration of comorbid disorders |
A study of alcoholics showed that traumatic experiences and symptoms associated with trauma are associated with a higher risk of relapse only in women.[44] The severity of trauma during childhood also proves to be a predictor of relapse in cocaine dependence only in women.[45] Dependent women who have received treatment for comorbid depression are more likely to be successful in drug treatment than depressed women without treatment. [46] To increase the likelihood of successful drug treatment, it is important to treat comorbid conditions in both women and men. |
|
Group therapies |
Men seem to benefit more from clear structures (such as in Alcoholics Anonymous). Women are more likely to need groups in which emotions are worked on and skills such as self-confidence or self-efficacy are practised in order for treatment to be successful. Gender-homogeneous groups are usually recommended for women, as they feel safer among women and relevant topics can be addressed better.[46] |
|
Cognitive behavioural therapy as a couple |
For women, social support is a factor that has a stronger influence on both the acceptance of therapy and the success of treatment than for men.[33] In addition, their partner's substance consumption has an increasing effect on their own consumption.[3] These aspects can be treated in women through joint cognitive behavioural therapy. Overall, the findings show that therapy together with the partner can be effective for the goal of reducing alcohol consumption or remaining abstinent. Individual sessions as a supplement increase this effect even more in women.[47] |
|
The female cycle |
The female cycle influences the subjective effect of substances as well as physiological reactions and withdrawal symptoms.[19] Particularly in women with nicotine addiction, attempts have been made to use the inhibitory effect of progesterone by initiating smoking cessation in the luteal phase (in which progesterone levels are highest). The findings are mixed: Some studies reported success in terms of a longer time to relapse, others found no effect of the cycle phase on treatment success, or withdrawal symptoms were actually lower in the follicular phase.[19] |
|
Impulsivity and inhibitory control functions |
Impulsivity (here especially the variant impulsive choice) is a predictor for treatment success for both sexes. Whereby higher values in women (who generally show lower levels) could be a risk factor for them to relapse more often than men.[6] Drug treatment should thus aim to reduce impulsivity or promote self-regulation in order to counteract substance use. In this context, women and men can benefit from different paradigms to strengthen their self-control.[48] For example, women seem to benefit better from earlier social contacts and from exercises concerning executive control (e.g. listening to music to improve self-control). |
|
Physical activity |
A review from 2015 shows that physical activity may be suitable for treating addictions. Initial experiments on rodents suggest this possibility.[6] A positive effect was observed more often in female and juvenile animals, and was also more pronounced.[3] |
Psychosocial factors
That social factors influence the gender-specific use of drugs is shown, for example, by the fact that the prevalence of substance abuse was higher among women from the 18th to the 19th century, men increasingly overtook women only from the 19th century onwards, and that nowadays there is a tendency for the sexes to equalise again for many substances.[6] There are indications that women react more sensitively to factors such as substance availability and social conventions. It is assumed that the stronger social condemnation of substance use among women acted as a protective factor for a long time, which is now less and less effective, so that the prevalences are becoming increasingly equalised.[31]
Women are much less likely than men to seek specific treatment for substance misuse, which has been linked to the following social factors, among others: Women are on average economically worse off, their education is on average lower, and they receive less social support if they are considered dependent. In order to overcome these barriers for women, services in terms of childcare, perinatal treatment or family services could be established, for example. The social image of addiction as a typically "male" disorder also urgently needs to change.[33]
Prevention
For both sexes, puberty is a critical phase for the onset of substance use and crucial for the later development of dependence, so that preventive programmes should start during or even before puberty.[3] Girls typically enter puberty earlier, which can also explain their earlier initial use. Therefore, prevention programmes might be necessary for girls earlier than for boys. Even if personality traits such as sensation seeking and impulsivity make both sexes more susceptible to substance use, special programmes should possibly be designed for boys. Both personality traits are more pronounced in boys than in girls from puberty onwards. This is thought to be one reason for the higher prevalence of substance dependence in males. Especially in girls, symptoms of depression or anxiety should be recognised and treated as early as possible, as these are strongly related to substance use.
In principle, prevention can start with the motivation to use substances. This varies between the sexes: men use drugs more for pleasure, women more to suppress negative affects (e.g. anxiety, depression or stress).[3] Accordingly, education is recommended for boys/men in which sensible alternatives for gaining pleasure are suggested and consequences of substance use are emphasised, while girls/women should increasingly be introduced to healthy, effective coping strategies.
Overall, it is necessary to create awareness in society that addiction also affects women and that addiction can have a different impact on women than on men. The aim should be that women (and men) take up drug therapies more often without fear of being judged.[33] In this way, the prevalence in both sexes could be reduced. Currently, there is still a lack of studies that take into account gender-specific effects of prevention programmes.[49]
Translation into patient care
Open research questions
So far, little attention has been paid to gender in addiction research, even though recently published reviews try to draw attention to this gap in the literature and to highlight possible gender-specific influences. In the future, it will become clear whether the reported gender differences regarding the course, the consequences or also the form of therapy will find their way into everyday clinical practice. Results suggest that both sexes would benefit from this.
Outlook
External Links
Literature
- Schmidt, C. O., Watzke, A. B., Schulz, A., Baumeister, S. E., Freyberger, H. J. & Grabe, H. J. (2013) Die Lebenszeitprävalenz psychischer Störungen in Vorpommern. Welchen Einfluss haben frühere psychische Auffälligkeiten auf die Survey-Teilnahme und Prävalenzschätzungen? Ergebnisse der SHIP-Studie. Psychiatrische Praxis, 40, 192–199
- Pabst, A., Kraus, L., De Matos, E. G., & Piontek, D. (2013). Substanzkonsum und substanzbezogene Störungen in Deutschland im Jahr 2012. Sucht, 59(6), 321–331.
- Kuhn, C. (2015). Emergence of sex differences in the development of substance use and abuse during adolescence. Pharmacology & Therapeutics, 153, 55–78.
- Fattore, L., & Melis, M. (2016). Sex differences in impulsive and compulsive behaviors: a focus on drug addiction. Addiction Biology, 21(5), 1043–1051.
- Shulman, E. P., Harden, K. P., Chein, J. M., & Steinberg, L. (2015). Sex differences in the developmental trajectories of impulse control and sensation-seeking from early adolescence to early adulthood. Journal of Youth and Adolescence, 44(1), 1–17.
- Carroll, M. E., & Smethells, J. R. (2016). Sex Differences in Behavioral Dyscontrol: Role in Drug Addiction and Novel Treatments. Frontiers in Psychiatry, 6:175.
- MacKillop, J., Amlung, M. T., Few, L. R., Ray, L. A., Sweet, L. H., & Munafò, M. R. (2011).
- Weafer, J., & de Wit, H. (2014). Sex differences in impulsive action and impulsive choice. Addictive Behaviors, 39(11), 1573–1579.
- Cross, C. P., Copping, L. T., & Campbell, A. (2011). Sexdifferences in impulsivity: a meta-analysis. Psychological Bulletin, 137(1), 97–130.
- Hosseini-Kamkar, N., & Morton, J. B. (2014). Sex differences in self-regulation: an evolutionary perspective. Frontiers in Neuroscience, 8:233.
- Medina, K. L., McQueeny, T., Nagel, B. J., Hanson, K. L., Schweinsburg, A. D., & Tapert, S. F. (2008). Prefrontal cortex volumes in adolescents with alcohol use disorders: unique gender effects. Alcoholism, Clinical and Experimental Research, 32(3), 386–394.
- Bazzett, T. J., & Becker, J. B. (1994). Sex differences in the rapid and acute effects of estrogen on striatal D2 dopamine receptor binding. Brain Research, 637(1-2), 163–172.
- Melis, M., De Felice, M., Lecca, S., Fattore, L., & Pistis, M. (2013). Sex-specific tonic 2-arachidonoylglycerol signaling at inhibitory inputs onto dopamine neurons of Lister Hooded rats. Frontiers in Integrative Neuroscience, 7:93.
- Cance, J. D., Ennett, S. T., Morgan-Lopez, A. A., Foshee, V. A., & Talley, A. E. (2013). Perceived pubertal timing and recent substance use among adolescents: a longitudinal perspective. Addiction (Abingdon, England), 108(10), 1845–1854.
- Clark, C. B., Perkins, A., McCullumsmith, C. B., Islam, M. A., Hanover, E. E., & Cropsey, K. L. (2012). Characteristics of victims of sexual abuse by gender and race in a community corrections population. Journal of Interpersonal Violence, 27(9), 1844–1861.
- Afifi, T. O., Henriksen, C. A., Asmundson, G. J. G., & Sareen, J. (2012). Childhood maltreatment and substance use disorders among men and women in a nationally representative sample. Canadian Journal of Psychiatry. Revue Canadienne de Psychiatrie, 57(11), 677–686.
- Hyman, S. M., Garcia, M., & Sinha, R. (2006). Gender specific associations between types of childhood maltreatment and the onset, escalation and severity of substance use in cocaine dependent adults. The American Journal of Drug and Alcohol Abuse, 32(4), 655–64.
- Lenz, B., Müller, C. P., Stoessel, C., Sperling, W., Biermann, T., Hillemacher, T., … Kornhuber, J. (2012). Sex hormone activity in alcohol addiction: Integrating organizational and activational effects. Progress in Neurobiology, 96(1), 136–163.
- Agabio, R., Campesi, I., Pisanu, C., Gessa, G. L., & Franconi, F. (2016). Sex differences in substance use disorders: focus on side effects. Addiction Biology, 21(5), 1030–1042.
- Erol, A., & Karpyak, V. M. (2015). Sex and gender-related differences in alcohol use and its consequences: Contemporary knowledge and future research considerations. Drug and Alcohol Dependence, 156, 1–13.
- Strickland, J. C., & Smith, M. A. (2014). The effects of social contact on drug use: Behavioral mechanisms controlling drug intake. Experimental and Clinical Psychopharmacology, 22(1), 23–34.
- Hostetler, C. M., Anacker, A. M. J., Loftis, J. M., & Ryabinin, A. E. (2012). Social housing and alcohol drinking in male-female pairs of prairie voles (Microtus ochrogaster). Psychopharmacology, 224(1), 121–132.
- Kirisci, L., Mezzich, A. C., Reynolds, M., Tarter, R. E., & Aytaclar, S. (2009). Prospective Study of the Association Between Neurobehavior Disinhibition and Peer Environment on Illegal Drug Use in Boys and Girls. The American Journal of Drug and Alcohol Abuse, 35(3), 145–150.
- Bisagno, V., & Cadet, J. L. (2014). Stress, sex, and addiction: potential roles of corticotropinreleasing factor, oxytocin, and arginine-vasopressin. Behavioural Pharmacology, 25(5-6), 445–457.
- Gillies, G. E., Virdee, K., McArthur, S., & Dalley, J. W. (2014). Sex-dependent diversity in ventral tegmental dopaminergic neurons and developmental programing: A molecular, cellular and behavioral analysis. Neuroscience, 282, 69–85.
- Becker, J. B., & Hu, M. (2008). Sex differences in drug abuse. Frontiers in Neuroendocrinology, 29(1), 36–47.
- Caldú, X., & Dreher, J.-C. (2007). Hormonal and genetic influences on processing reward and social information. Annals of the New York Academy of Sciences, 1118(1), 43–73.
- Greenfield, S. F., Back, S. E., Lawson, K., & Brady, K. T. (2010). Substance Abuse in Women. Psychiatric Clinics of North America, 33(2), 339–355.
- Bobzean, S. A. M., DeNobrega, A. K., & Perrotti, L. I. (2014). Sexdifferences in the neurobiology of drug addiction. Experimental Neurology, 259, 64–74.
- Becker, J. B., McClellan, M., & Reed, B. G. (2016). Sociocultural context for sex differences in addiction. Addiction Biology, 21(5), 1052–1059.
- Becker, J. B., McClellan, M., & Reed, B. G. (2016). Sociocultural context for sex differences in addiction. Addiction Biology, 21(5), 1052–1059.
- Weißbach L. & Stiehler, M. Männergesundheitsbericht 2013: Im Fokus: Psychische Gesundheit. Bern: Hans Huber; 2013
- Greenfield, S. F., Brooks, A. J., Gordon, S. M., Green, C. A., Kropp, F., McHugh, R. K., … Miele, G. M. (2007). Substance abuse treatment entry, retention, and outcome in women: a review of the literature. Drug and Alcohol Dependence, 86(1), 1–21.
- Agabio, R., Pani, P. P., Preti, A., Gessa, G. L., & Franconi, F. (2016). Efficacy of Medications Approved for the Treatment of Alcohol Dependence and Alcohol Withdrawal Syndrome in Female Patients: A Descriptive Review. European Addiction Research, 22(1), 1–16.
- Greenfield, S. F., Pettinati, H. M., O’Malley, S., Randall, P. K., & Randall, C. L. (2010). Gender differences in alcohol treatment: an analysis of outcome from the COMBINE study. Alcoholism, Clinical and Experimental Research, 34(10), 1803–1812.
- Martinotti, G., Di Nicola, M., Tedeschi, D., Andreoli, S., Reina, D., Pomponi, M., … Janiri, L. (2010). Pregabalin versus naltrexone in alcohol dependence: a randomised, double-blind, comparison trial. Journal of Psychopharmacology, 24(9), 1367–1374.
- Graziani, M., Nencini, P., & Nisticò, R. (2014). Genders and the concurrent use of cocaine and alcohol: Pharmacological aspects. Pharmacological Research, 87, 60–70.
- Pettinati, H. M., Kampman, K. M., Lynch, K. G., Suh, J. J., Dackis, C. A., Oslin, D. W., & O’Brien, C. P. (2008). Gender differences with high-dose naltrexone in patients with co-occurring cocaine and alcohol dependence. Journal of Substance Abuse Treatment, 34(4), 378–390.
- Roche, D. J. O., & King, A. C. (2015). Sex differences in acute hormonal and subjective response to naltrexone: The impact of menstrual cycle phase. Psychoneuroendocrinology, 52, 59–71.
- Garbutt, J. C., Kranzler, H. R., O’Malley, S. S., Gastfriend, D. R., Pettinati, H. M., Silverman, B. L.,… Vivitrex Study Group. (2005). Efficacy and tolerability of long-acting injectable naltrexone for alcohol dependence: a randomized controlled trial. JAMA, 293(13), 1617–1625.
- Minami, H., Kahler, C. W., Bloom, E. L., Prince, M. A., Abrantes, A. M., Strong, D. R., … Brown, R. A. (2014). Effects of sequential fluoxetine and gender on prequit depressive symptoms, affect, craving, and quit day abstinence in smokers with elevated depressive symptoms: A growth curve modeling approach. Experimental and Clinical Psychopharmacology, 22(5), 392–406.
- Yonkers, K. A., Forray, A., Nich, C., Carroll, K. M., Hine, C., Merry, B. C., … Sofuoglu, M. (2014). Progesterone for the reduction of cocaine use in post-partum women with a cocaine use disorder: a randomised, double-blind, placebo-controlled, pilot study. The Lancet Psychiatry, 1(5), 360–367.
- Sofuoglu, M., Mitchell, E., & Kosten, T. R. (2004). Effects of progesterone treatment on cocaine responses in male and female cocaine users. Pharmacology, Biochemistry, and Behavior, 78(4), 699–705.
- Heffner, J. L., Blom, T. J., & Anthenelli, R. M. (2011). Gender Differences in Trauma History and Symptoms as Predictors of Relapse to Alcohol and Drug Use. The American Journal on Addictions,20(4), 307–311.
- Hyman, S. M., Paliwal, P., Chaplin, T. M., Mazure, C. M., Rounsaville, B. J., & Sinha, R. (2008). Severity of childhood trauma is predictive of cocaine relapse outcomes in women but not men. Drug and Alcohol Dependence, 92(1-3), 208–216.
- Zenker, C. (2005). Sucht und Gender. Bundesgesundheitsblatt Gesundheitsforsch. Gesundheitsschutz, 48(4), 469–476.
- McCrady, B. S., Epstein, E. E., Hallgren, K. A., Cook, S., & Jensen, N. K. (2016). Women with alcohol dependence: A randomized trial of couple versus individual plus couple therapy. Psychology of Addictive Behaviors, 30(3), 287–299.
- Mansouri, F. A., Fehring, D. J., Gaillard, A., Jaberzadeh, S., & Parkington, H. (2016). Sex dependency of inhibitory control functions. Biology of Sex Differences, 7(1), 11.
UNODC. (2016). Guidelines on drug prevention and treatment for girls and women. Retrieved from https://www.unodc.org/documents/drug-prevention-and-treatment/unodc_2016_drug_prevention_and_treatment_for_girls_and_women_E.pdf
License
This article is published under the Creative Commons License. The full license content can be retrieved here: https://creativecommons.org/licenses/by/3.0/legalcode
Autoren
Last changed: 2022-04-26 10:59:02